Zinc and Copper functions and the implications of a high copper to zinc ratio in the body
The human body is made up of both organic and non-organic matter. Almost 99% of the body is made up of six elements, these are oxygen, carbon, hydrogen, nitrogen, calcium and phosphorous. (1) The remaining amount is made up of trace minerals. Trace minerals are a range of minerals that are needed in the body in small amounts, some of these minerals include iron, iodine, selenium, fluoride, chromium, zinc and copper. Although the trace minerals are in very small quantities in the body, they are extremely important and imbalances of trace minerals can cause havoc on a person’s health. Trace minerals activate many enzymes responsible for a range of biochemical functions and are an important part of the overall functioning of the body.
Trace mineral deficiencies are known to be involved in many disorders of the endocrine, cardiovascular, gastrointestinal, musculoskeletal, neurological and immune systems. (2) It is not just absolute deficiencies that can cause problems but also the ratio of a particular mineral to another particular mineral in the body. Some of the significant ratios in the body are calcium/phosphorous (Ca/P), sodium to potassium (Na/K), zinc to copper (Zn/Cu), sodium to magnesium (Na/Mg) and iron to copper (Fe/Cu). (3) In this review, we are going to analyse the function of both zinc and copper and the effects of an imbalance between these two trace minerals.
Zinc
Zinc was discovered as an essential trace mineral for the growth of living organisms in 1869 for plants, in 1934 for animals and in 1961 for humans. (4) Zinc is the second most common trace mineral in the body after iron and is present in every living cell. There is around 2-4g of zinc in the body at any one time, the highest concentrations are found in the eye and prostate with other concentrated areas being in the brain, muscle, skin, bones, kidney and liver. Zinc is heavily involved in cellular metabolism and is required for more than 200 enzyme reactions within the body. (5) Zinc is heavily involved in wound healing, protein synthesis, DNA synthesis and cell division. It supports normal growth and development during pregnancy, childhood and adolescence and is required for adequate taste and smell throughout life. Zinc is also understood to be antimicrobial, have antioxidant properties and can help in protecting against accelerated aging and speed up the wound healing process. (6)
Zinc deficiency signs can vary between individuals, this can depend upon whether there is an absolute or relative deficiency. An absolute deficiency is when there is increased requirements from the body, a lack of intake or mineral loss through things such as stress, illness, increased thyroid activity, medications or heavy metal accumulation. A relative deficiency is where zinc is lost from tissue storage sites due to in imbalance with another antagonistic mineral such as copper or cadmium. (7) Zinc deficiency signs can be often seen in the skin and with white spots on the nails.
Zinc deficiency is common is clients with skin disease such as lupus erythematosus (SLE), psoriasis and scleroderma. Stretch marks are also a sign of a low level of zinc and zinc deficiency can contribute to skin blemishes, rashes and the slow healing of wounds. (8) Other disease states that often accompany a low level of zinc include sickle cell anaemia, diabetes, anorexia, menstrual irregularities and recurrent viruses and infections. Insulin storage requires adequate zinc levels, copper accumulation can be the cause of low zinc in issues with anorexia and menstrual irregularities. Zinc is one of minerals that is essential to maintain the hormone levels of progesterone, estrogen and testosterone while copper is associated to estrogen. An imbalance in this ratio can be a direct cause of PMS and other menstrual irregularities. (7, 9)
A low zinc level is also involved with sleep disturbances and depression, zinc is involved in both the production of melatonin and serotonin. The following chart shows the conversion of L-Tryptophan to both serotonin and melatonin, requiring zinc and other nutrients as part of the conversion process. (10)

L-Tryptophan
- Requires Folate, calcium, iron and vitamin B3
5-Hydroxytryptophan
- Requires vitamin B6, zinc, magnesium and Vit C
Serotonin
- Happy chemical
Melatonin
- Sleep chemical
Figure 1. The biodiversity of essentials that require zinc (10)
Zinc is essential in the production of stomach acid, a deficiency in zinc can cause low stomach acid and the inability to effectively absorb zinc and other nutrients from food creating multiple deficiencies throughout the body including a further zinc deficiency.
The established recommended daily intake (RDI) of zinc is 15mg or 0.2mg/kg. Foods high in zinc include beef, bilberry, capsicum, egg yolk, ginger, liver, milk, lamb, oysters, sunflower and pumpkin seeds, seafood and whole grains. (11) Despite zinc being readily available from food, approximately half of the population is at a risk of zinc deficiency. (11) Farming practices, food quality, nutrient imbalances and poor absorption are all major contributing factors to low levels of zinc in society. (10, 11)
Copper
Copper is an essential trace mineral required for many chemical, cellular and enzymatic functions within the body. Copper is found most abundantly in the brain and liver, with other accumulation sites being in the heart, lungs, kidneys, pancreas, muscles and bones. Copper plays a major role in oxygen transport, mental and cognitive function, immunity, energy production, neurotransmitter production and bone and nerve tissue maintenance. (12) A deficiency or excess in copper will disrupt the function of copper dependent enzyme reactions in the body. Copper is an essential factor in cellular enzymes functions as well as vital to cell membrane structure including cytochrome c oxidase, superoxide dismutase, dopamine B-hydroxylase, lysol oxidase, tyrosinase and monoamine oxidase. (13)
Copper excess is far more common than copper deficiency, particularly in Australia and America. Highly farmed areas often have high copper and low zinc levels that contribute greatly to the problem. Drinking water, copper water pipes, copper added to animal feed lots and pesticides sprayed on fruits and vegetables are all ways that copper can get into the food chain. (14) Copper can accumulate in the body at different speeds depending on a person’s metabolic activity, the slower the metabolic rate the higher the copper retention. Copper excess has also been linked to prescribed oestrogens and xenoestrogen. A range of studies have been undertaken and found that copper accumulation increases with the intake of these estrogens. The estrogens can come from the oral contraception pill (OCP), hormone replacement, pesticides, plastics, petroleum based products and antibiotics given to animals that enter the food chain. (10, 15, 16)
Conditions associated with excess copper include premenstrual syndrome (PMS), gallstones, viral infections, yeast and fungus overgrowth, scoliosis and a range of mental and behavioural disorders. With PMS, as the copper levels rise within the body so does estrogen, causing a hormonal imbalance and contributing to PMS. (17) Viral, yeast and fungal infections all thrive in a high copper environment. Viral infections lower a person’s metabolic rate promoting the retention of copper. (7) Excess Copper is associated with many mental conditions, it can contribute to the symptoms of depression, panic attack, anxiety, fearfulness, excessive anger and schizophrenia. (18, 19)
The established RDI for copper is 1-3mg per day. Foods high in copper include beef liver, sunflower seeds, lentils, almonds, chocolate, asparagus, avocado and mushrooms. (11) Many vegetarians can become copper toxic through the large amounts high copper foods commonly eaten by vegetarians and the absence of meat that has a high zinc content to offset the copper. This outlines the importance of the zinc to copper ratio.
Zinc to Copper Ratio
The levels of zinc and copper are important in the body but just as important is the zinc/copper ratio. The ideal ratio between zinc and copper is 8:1 with an acceptable range from 4 to 12. (20) Both zinc and copper have a range of other nutrients that either antagonise or promote absorption of these nutrients. Vitamins antagonistic to copper are B6, A, C, B3 and B5. Minerals antagonistic to copper (Cu) are molybdenum (Mo), Cadmium (Cd), Mercury (Hg), zinc (Zn), Iron (Fe), Lead (Pb), Potassium (K), and Phosphorous (P). Synergistic nutrients include vitamin D, B1, B12, C and folic acid.
(21) The copper wheels show the antagonistic relationships between Cu and other vitamins and minerals, the wheels show Zn and vitamin C as the most antagonistic mineral and vitamin respectively.
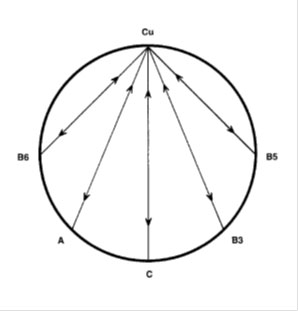
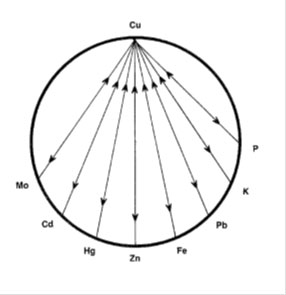
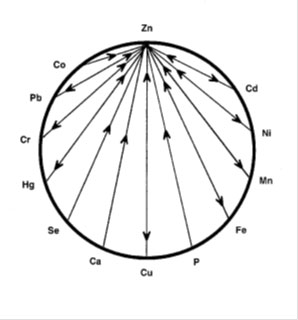
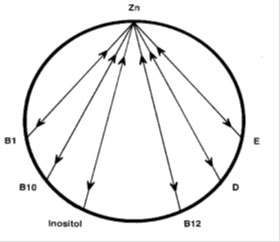
The zinc wheels show the vitamins and minerals that are antagonistic to zinc. High intakes of any of these antagonists can result in low or deficient zinc levels.
Both copper and zinc are regulated by metallothionein (MT), a low molecular weight cysteine-rich, metal binding protein originates that is comprised of 61 amino acid units. (23) If this protein isn’t functioning properly within the body, abnormal nutrient levels of copper, zinc, manganese, cadmium, lead and mercury can result. MT activity can be significantly reduced through either zinc deficiency or elevated oxidative stress.
As both zinc and copper are regarded as neurotransmitters and are high in concentrations in the brain, a high copper to zinc ratio is associated with many behavioural disorders such as attention deficit disorder (ADD), depression, paranoid schizophrenia, bipolar disorder and violent behaviour. (19) Nutrient therapy can be effective in normalising copper and zinc levels and in turn can be effective in balancing dopamine and norepinephrine levels in the brain, drastically improving and normalising mental function.
Studies
There have been a number of studies conducted with regards to the balancing of zinc and copper in the body and outlining the profound effects that can be achieved in both mental complaints, physical complaints and oxidative stress.
After over 20 years of research observations in patients with depressed zinc levels and elevated copper, William J Walsh conducted a study in 1996 of 49 young males between the ages of 3 and 20 with reported behaviour disorders and an additional 17 males in the control group with no previously reported behavioural disorders, the mean age of the group was 10 years old. The objective of the study was assess whether a high Cu/Zn ratio contributed to violent behaviours in young adults. The mean Cu/Zn serum ratio in the assaultive subjects was 1.40 compared to the control group of just 1.02. Results were also categorised into intensities of behaviour with the highest aggravated individuals having the highest Cu/Zn ratios. The findings of this study suggest a correlation between an elevated Cu/Zn ratio and violence prone behaviour. In addition, four separate outcome studies have shown a significant improvement in behaviour after balancing the Cu/Zn ratio. With the biochemical nutrient therapy of 207 patients with diagnosed behavioural therapy, 76% of the group completed the nutrient therapy and a total of 88% of these patients exhibited a reduced frequency of destructive behaviour and 53% totally eliminated the behaviour. Therapy was individualised based on blood work and presenting symptoms, it consisted of a range of supplements including methionine, zinc, vitamin B6, magnesium, pyridoxine-5-phosphate (P5P) and vitamins C and E. (25)
Another study in regards to behavioural disorders was conducted on 230 children with autism spectrum disorders to research the plasma zinc/serum copper ratio as a biomarker for the condition. The entire cohort’s mean zinc level was 77.2 μg/dl, mean copper level was 131.5μg/dl and Zn/Cu ratio was 0.608. The cut-off for the lowest 2.5% of healthy children surveyed was 0.7, indicating that a low Zn/Cu ratio may be a biomarker of heavy metal toxicity, particularly mercury, in children with autism spectrum disorders. (26)
Another study assessed the Cu/Zn ratio and systemic oxidant load with the effect on aging and age related degenerative disease. The study measured the blood content of copper, zinc and ceruloplasmin as well as lipid peroxides in 81 healthy and 62 disabled individuals in their 90’s who were affected by chronic degenerative disease. The serum Cu/Zn ratio and ceruloplasmin were all significantly higher in the elderly with the parameters being significantly higher in the disabled elderly over the elderly without chronic degenerative disease. In addition, those with chronic degenerative disease had depressed zinc and elevated copper levels. The study concluded that there is a strict relationship between copper and zinc and the systemic oxidation on the body. Furthermore, advanced age chronic degenerative diseases are associated with a high Cu/Zn ratio and systemic oxidative stress and the Cu/Zn ratio may serve as useful predictive biomarkers for poor health in the elderly. (27)
An additional study also measured the plasma Cu/Zn ratio as an inflammatory / nutritional biomarker as predictor of all-cause mortality in elderly population. The association between plasma Cu/Zn and inflammatory (CRP, ESR, IL-6) or nutritional (albumin, BMI) markers was studied in 498 elderly Italian subjects. Blood samples were also taken from a control group of 164 healthy people aged between 20 and 60. A prospective follow-up study of mortality by age-related diseases was performed with 218, 70+ year old people after a duration of 3.5 years. Subjects with cardiovascular disease all had higher plasma Cu/Zn ratios than those without, mainly due to increased plasma Cu. In other subjects, most of the age related decline appeared to be from a progressive decline in plasma Zn. The study highlights the importance of the levels of zinc in the body and the detrimental health effects when the Cu/Zn ratio is high. (28)
The effects of a high Cu/Zn ratio may also play a part in the formation of cancer. A study measuring plasma Cu and Zn was conducted in 1978 on 126 patients, 54 with carcinomas, 40 of those with carcinomas of digestive organs, 37 with benign lesions and 35 subjects without disease. The plasma Cu level increased while the plasma Zn level decreased significantly in the group with the carcinoma of the digestive organs. The Cu/Zn ratio was significantly higher than in the non-disease group. From this study it was concluded that a high Cu/Zn ratio may be valuable in estimating the extent of the carcinoma and determining prognosis of cancer patients. (29)
Further study of a group of 55 breast cancer patients, 20 with benign breast disease and 35 with breast cancer and an additional 30 control subjects were studied. The mean serum copper levels were higher in breast cancer than in benign disease and the controls (167μg/dl vs 117.6μg/dl). The serum copper levels were also significantly higher in the more advanced breast cancer patients over those with early breast cancer (177.9μg/dl vs 130.4μg/dl). Mean serum zinc levels were only lowered in advanced breast cancer patients when compared to controls. The Cu/Zn ratio was increased in breast cancer patients but not in patients with benign breast disease. It was found that the serum copper levels and the Cu/Zn ratio may be used as a biochemical marker in breast cancer patients. (30)
A study of 60 patients with psoriasis were studied in 2005 and their serum Cu and Zn levels were tested and compared with 20 similar aged healthy controls. The mean age of patients was 31.6 with the duration of psoriasis lasting from one month to 18 years. Serum Zn levels in the healthy controls ranged from 98-118 μg/dl (mean 106.6 μg/dl) while the psoriasis patients range was 58-96 μg/dl (mean 74.28 μg/dl). Serum copper levels in the controls ranged from 102-126 μg/dl (mean 115.14 μg/dl) while in the psoriasis patients it ranged from 122-160 μg/dl (mean 142.78 μg/dl). The difference between the two groups was significant with psoriasis patients having significantly lower zinc and higher copper than the healthy control group. The Cu/Zn ratio was also significantly higher in the psoriasis patients, being 1.92 when compared to the control of just 1.08. The study concluded that significantly lower zinc levels and higher copper levels may play a role in the disease progression of psoriasis and correcting this imbalance may assist in the treatment of psoriasis. (31)
Conclusion
The role of both zinc and copper in the body are of the utmost importance. Zinc is essential for healthy immune function, the reproductive system, normal growth and development and a range of other functions within the body. It is also a major mineral for hormone production and is a copper antagonist. Copper is required for a healthy nervous system, bones, joints, skin, blood vessels, mood regulation and myelin sheath formation. When levels become too high, particularly when zinc is low, toxic effects on the body can be observed.
Zinc is commonly low when copper is high as copper has a strong antagonistic effect on zinc. As both copper and zinc antagonise each other, health issues can occur if the ratios are out of balance either way. Low copper and high zinc can also cause a wide range of adverse health effects. There are a number of studies that show the detrimental effects on the mental state of patients and that a high Cu/Zn ratio. This imbalance appears to play a contributing role in many mental diseases including schizophrenia, anxiety, depression and ADD.
The effects of excess copper and low zinc also appear to promote rapid oxidation in the body. Studies found that elderly patients with degenerative disease had significantly higher Cu/Zn ratios than elderly individuals without chronic degenerative disease. Cancer formation also can be found within studied patients that had high Cu/Zn ratios again suggesting the oxidative and biochemical disruption such as state has on the body. It would be wise to consider the possibility of a high copper and low zinc scenario that may be having a detrimental effect on many clients that present with a wide range of conditions as outlined in this review.
Jarrod Cooper - 2016
References
- Minerals: Their Functions and Sources-Topic Overview [Internet]. WebMD. 2016 [cited 10 November 2016]. Available from: http://www.webmd.com/vitamins-and-supplements/tc/minerals-their-functions-and-sources-topic-overview
- Prasad CGowda N. Importance of trace minerals and relevance of their supplementation in tropical animal feeding system: A review [Internet]. Epubs.icar.org.in. 2016 [cited 10 November 2016]. Available from:http://epubs.icar.org.in/ejournal/index.php/IJAnS/article/view/9668
- ARL : Basic Ratios and Their Meaning [Internet]. Arltma.com. 2016 [cited 10 November 2016]. Available from: http://www.arltma.com/Articles/RatiosDoc.htm
- Zinc - Trace elements - Nutrients - Home [Internet]. Nutri-facts.org. 2016 [cited 10 November 2016]. Available from: http://www.nutri-facts.org/en_US/nutrients/trace-elements/zinc.html
- HH S. Understanding zinc: recent observations and interpretations. - PubMed - NCBI [Internet]. Ncbi.nlm.nih.gov. 2016 [cited 10 November 2016]. Available from: https://www.ncbi.nlm.nih.gov/pubmed/8083574
- N FE M. Zinc, human diseases and aging. [Internet]. Europepmc.org. 2016 [cited 10 November 2016]. Available from: http://europepmc.org/abstract/MED/7548268
- Watts D. Trace elements and other essential nutrients. 1st ed. [Place of publication not identified]: [publisher not identified]; 1995.
- Zinc [Internet]. Linus Pauling Institute. 2016 [cited 10 November 2016]. Available from: http://lpi.oregonstate.edu/mic/minerals/zinc
- The ARL Newsletter [Internet]. Drlwilson.com. 2016 [cited 10 November 2016]. Available from: http://drlwilson.com/Articles/ZINC.htm
- Tabrizian I. Nutritional medicine. 1st ed. [Yokine, WA]: NRS Pub.; 2002.
- Osiecki H. The Nutrient Bible. 9th ed. 2005.
- Cognitive Function [Internet]. Linus Pauling Institute. 2016 [cited 11 November 2016]. Available from: http://lpi.oregonstate.edu/mic/health-disease/cognitive-function
- Hordyjewska A, Popiołek Ł, Kocot J. The many “faces” of copper in medicine and treatment. 2016. Available from : http://link.springer.com/article/10.1007/s10534-014-9736-5
- Wuana Rokieimen F. Heavy Metals in Contaminated Soils: A Review of Sources, Chemistry, Risks and Best Available Strategies for Remediation. 2016. Available from : https://www.hindawi.com/journals/isrn/2011/402647/
- R M. Effect of estrogen on serum and tissue levels of copper and zinc. - PubMed - NCBI [Internet]. Ncbi.nlm.nih.gov. 2016 [cited 15 November 2016]. Available from: https://www.ncbi.nlm.nih.gov/pubmed/2626984
- COPPER TOXICITY SYNDROME [Internet]. Drlwilson.com. 2016 [cited 15 November 2016]. Available from: http://www.drlwilson.com/articles/copper_toxicity_syndrome.htm
- ARL : New Knowledge About Premenstrual Syndrome [Internet]. Arltma.com. 2016 [cited 15 November 2016]. Available from: http://www.arltma.com/Articles/PMSDoc.htm
- COPPER TOXICITY SYNDROME [Internet]. Drlwilson.com. 2016 [cited 15 November 2016]. Available from: http://www.drlwilson.com/articles/copper_toxicity_syndrome.htm
- Walsh W. Nutrient power. 1st ed. New York, NY: Skyhorse Pub.; 2012.
- Watts D. HTMA MIneral Ratios [Internet]. 1st ed. 2010 [cited 15 November 2016]. Available from: http://t.traceelements.com/Docs/newsletternov-dec2010.pdf
- Copper Wheels [Internet]. 1st ed. 2016 [cited 15 November 2016]. Available from: http://www.traceelements.com/docs/Copper%20Wheels.pdf
- Zinc Wheels [Internet]. 1st ed. 2016 [cited 15 November 2016]. Available from: http://www.traceelements.com/docs/Zinc%20Wheels.pdf
- Zinc-Copper Imbalances [Internet]. Drkaslow.com. 2016 [cited 15 November 2016]. Available from: http://www.drkaslow.com/html/zinc-copper_imbalances.html
- Walsh W. Elevated Blood Copper/Zinc Ratios in Assaultive Young Males [Internet]. 1st ed. 1996 [cited 15 November 2016]. Available from: http://www.hriptc.org/pdfs/Elevated%20Blood%20Cu%20and%20Zn%20Ratios%20in%20Assaultive%20Young%20Males.pdf
- Walsh W. Reduced violent behavior following biochemical therapy [Internet]. Walsh Institute. 2016 [cited 15 November 2016]. Available from: http://www.walshinstitute.org/uploads/1/7/9/9/17997321/walshbehaviorpaper6-04_1_.pdf
- The plasma zinc/serum copper ratio as a biomarker in children with autism spectrum disorders: Biomarkers: Vol 14, No 3 [Internet]. Tandfonline.com. 2016 [cited 15 November 2016]. Available from: http://www.tandfonline.com/doi/full/10.1080/13547500902783747?scroll=top&needAccess=true
- Copper/zinc ratio and systemic oxidant load: effect of aging and aging-related degenerative diseases [Internet]. Sciencedirect.com. 2016 [cited 15 November 2016]. Available from: http://www.sciencedirect.com/science/article/pii/S0891584998001099
- Malavolta M, Giacconi R, Piacenza F, Santarelli L, Cipriano C, Costarelli L et al. Plasma copper/zinc ratio: an inflammatory/nutritional biomarker as predictor of all-cause mortality in elderly population. 2016. Available from : https://www.ncbi.nlm.nih.gov/pubmed/19821050
- Inutsuka SAraki S. Plasma copper and zinc levels in patients with malignant tumors of digestive organs. Clinical evaluation of the Cu/Zn ratio [Internet]. Onlinelibrary.wiley.com. 2016 [cited 15 November 2016]. Available from: http://onlinelibrary.wiley.com/doi/10.1002/1097-0142(197808)42:2%3C626::AID-CNCR2820420232%3E3.0.CO;2-F/abstract
- Gupta S, Shukla V, Vaidya M, Roy S, Gupta S. Serum trace elements and Cu/Zn ratio in breast cancer patients. 2016. Available from : https://www.ncbi.nlm.nih.gov/pubmed/2011029
- Nigam P. Serum zinc and copper levels and Cu: Zn ratio in psoriasis. 2016. Available from http://www.ijdvl.com/article.asp?issn=0378-6323;year=2005;volume=71;issue=3;spage=205;epage=206;aulast=Nigam
[widget id="text-4"]
